Temperature and Resistance of Electronic Materials
Objective: The object of this lab is to show how temperature affects the conductivity (resistance) in various electrical materials and devices.
Review of Scientific Principles:
Heat: As heat is applied to a crystalline solid, we say "it gets hotter"; meaning the temperature increases. On the atomic level, the kinetic energy of the atoms has increased which means the atoms are moving faster. However, in a crystalline solid, the atomic movement is limited to vibration around stable lattice positions. As the temperature increases, the atoms vibrate at a greater amplitude and move farther from their stable lattice positions. This motion has a negative effect on the ability of the material to conduct an electric current, causing it to have a greater electrical resistance.
Metals: In a metal, the valence electrons are thought of as being shared by all the positive ions. Therefore, the electrons are free to move throughout the crystalline lattice. The electrons move randomly throughout the crystal, until an electric field is applied to the material. Then the electric field forces the electrons to move in a direction opposite to the field. Actually, the electrons still move somewhat randomly, but with a superimposed "drift". This produces current. As the temperature increases, the positive ions in the crystal vibrate more, and more collisions occur between the valence electrons and the vibrating ions. These collisions hinder the "drift" motion of the valence electrons, thus reducing the current. In summary, for a metal, an increase in temperature causes an increase in resistance.
Semiconductors: In a semiconductor, at 0 K, valence electrons are in filled energy levels (bonds are formed by electron pairs filling the energy levels). They do not respond to an applied electric field to produce current flow. In the presence of an electric field, the electron motion is still random; no net motion in one direction occurs (no current flows). These filled energy levels, which the valence electrons occupy, form the valence band. In order for current to flow, electrons must move from the filled valence band to the empty conduction band. To make this move requires energy, which can be in the form of heat. (Important: the electrons do not move from a "place" in the crystal called the valence band to another "place" called the conduction band. The electrons have the energy associated with the valence band and acquire enough energy to have the energy associated with the conduction band. An energy change occurs, not a position change.) At room temperature, many electrons will have the energy needed to jump to the conduction band. As one electron moves out of the valence band and into the conduction band, a hole (vacancy) is produced in the valence band. Both the electrons in the conduction band and the corresponding holes in the valence band are considered charge carriers. When an electric field is applied to the material, these electrons and holes "drift". The electrons in the conduction band drift in the direction opposite to the applied field, and the holes drift in the same direction as the applied field. Thus, current is produced. As the temperature of the material is increased, more valence electrons acquire sufficient energy to move to the conduction band (producing holes), so more current flows. It is still true that as the temperature is increased, the atoms vibrate more and cause more collisions with the drifting electrons. However, this opposing effect is negligible, compared to the increase in charge carriers.
Applications:
Different types of materials respond differently to temperature changes. A computer engineer designing a circuit must be able to predict if the conductivity of each material in the device will be within an acceptable range over the expected temperature range of operation of the device.
Time: One hour
Materials and Supplies:
heat source for boiling water (hot plate preferred)
5 beakers for water baths
thermometer
choke coils or resistance spools
germanium diodes
thermistors
light emitting diodes (LEDs)
carbon resistors
glass rod (5 cm)
2 digital multimeters or a voltmeter and milliammeter
wire connectors with alligator clips
power supply (0 to 12 volts DC)
General Safety Guidelines:
* The heat source could cause burns. Exercise caution.
* Be careful of electrical shock.
* Handle meters and samples with care.
* Wear safety glasses.
Procedure:
1. Set up five water baths of about 100-200 ml of water in beakers at the following
temperatures: boiling, 75 ° C, 50 ° C, 25 ° C, and ice water.
2. Measure the temperature of each bath with a thermometer, thermistor, or
thermocouple.
3. For measuring the resistance of the device (choke or resistance coil) set up the
multimeter to read ohms and connect as in the following diagram.
4. Carefully holding on to the lead wires so as not to burn the fingers, immerse the
coil into the boiling water bath, until a stable value is received (for about one
minute) and record the resistance in the data table.
5. Follow the same procedure in the 75 °, 50 °, 25 ° and ice water.
6. Remove the coil and attach another device to the meter, following the same
procedure for measuring the resistance.
Data and Analysis:
| CONDITION | TEMP. C | CHOKE COIL | GERMANIUM DIODE |
THERMISTOR | |||||
|---|---|---|---|---|---|---|---|---|---|
| ° | R | ° | R | ° | R | ° | R | ||
| Boiling water | 100 | ||||||||
| Hot water | 75 | ||||||||
| Warm water | 50 | ||||||||
| Room temp | 25 | ||||||||
| Ice water | 0 | ||||||||
Questions:
1. Which samples had a change in resistance as the temperature increased? What
direction was that change?
2. As their temperatures increased, what happened to the resistance of the
conductors, the semiconductors? Does the change seem to be linear?
3. Did any of the examples not follow the general guidelines explained in the
introduction to the lab? Explain.
4. Describe the motion of the atoms or ions in a crystalline solid as the temperature
increases.
5. What causes electrons to "drift"?
6. Describe the electron motion while current is flowing.
7. Explain how increasing the temperature of a semiconductor decreases the
resistance.
8. Explain how increasing the temperature of a metal increases its resistance.
Extension:
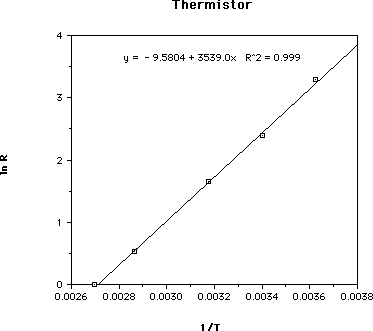
For the thermistor, plot 1/T (K-1) on the x-axis and ln R (natural log of the value of the resistance in ohms). This graph is a straight line. The equation of this line is:
ln R = (Egap / 2k) x 1/T + ln Ro
where:
k = 8.62 x 10-5 eV/K (Boltzman's constant)
Egap = band gap energy (the difference in energy between the conduction and valence bands) in electron volts.
Determine the slope of the line from the graph. (Egap / 2k) = the slope, from the equation. Solve this equation for Egap.
Teacher Notes:
*Teacher preparation time is about 30 minutes.
* Resistance coils could be used instead of a choke coil. (Short pieces of wire do not show enough resistance.)
* Other kinds of wire besides copper could be tried.
* Connector leads to devices could be extended by soldering on short lengths of wire.
* The carbon device should lose only a small percentage of its room temperature resistance, but semiconductor devices should go up appreciably at low temperature.
* As an example of a nonconductor , a length of glass rod could be tested.
* Use a Type K thermocouple for lower temperatures in conjunction with some digital multimeters.
* Thermistor probes are available form Vernier.
* The teacher should demonstrate proper hookups of meters and devices.
* Diode results work best if the temperature is taken from the boiling water and the ice water.
* For the extension activity, using the sample data, the value of Egap= 0.6 eV.
Answers to Questions:
1. All the devices changed their resistance as the temperature changed. The
resistance of the choke coil (which is copper wire) increased as the temperature
increased. The resistance of the diode and the thermistor (which are made of
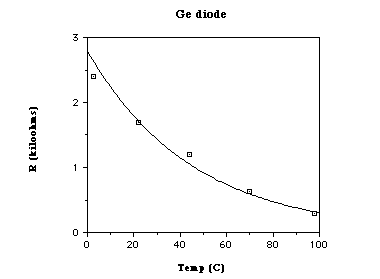
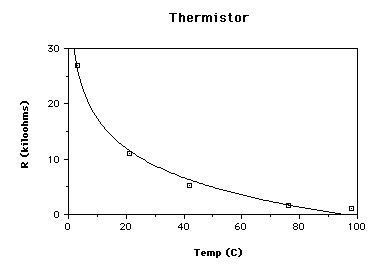
semiconductor material) decreased as the temperature increased.
2. The resistance of the conductor increased linearly. The resistance of the
semiconductor decreased, but not linearly.
3. Student answers will vary. The devices do react as theoretically predicted.
4. As the temperature increases, the atoms or ions vibrate with greater amplitude
around their stable lattice positions.
5. When an electric field is applied, the electrons are forced to drift .
6. The electrons are moving randomly and drifting in the opposite direction of the
applied electric field.
7. As the temperature increases, more electrons have the energy needed to move to
the conduction band (more charge carriers means more current).
8. Greater amplitude of vibration of the ions in the lattice cause more collisions with
the valence electrons, which decreases the drift velocity.
| Condition | Temp ° C | Choke Coil | Germanium Diode | Thermistor | |||||
|---|---|---|---|---|---|---|---|---|---|
| ° | R | ° | R k | ° | R k | ° | R k | ||
| Boiling Water | 100 | 97 | 44 | 98 | 0.29 | 98 | 1.0 | ||
| Hot Water | 75 | 77 | 41 | 70 | 0.63 | 76 | 1.7 | ||
| Warm Water | 50 | 51 | 37 | 44 | 1.2 | 42 | 5.2 | ||
| Room Temp | 25 | 22 | 33 | 22 | 1.7 | 21 | 11 | ||
| Ice Water | 0 | 3.5 | 31 | 2.8 | 2.4 | 3 | 27 | ||