Demonstration 3
The Formation of the Wonder Polymer
The Condensation Polymerization Reaction Used in the Creation of Nylon 6-10
Objective: The objective of this demonstration is to show the formation of a condensation polymer.
Review of Scientific Principles:
The word "nylon" is used to represent synthetic polyamides. The various nylons are described by a numbering system that indicates the number of carbon atoms in the monomer chains. Nylons from diamines and dicarboxylic acids are designated by two numbers, the first representing the diamine and the second the dicarboxylic acid. Thus nylon 6-10 is formed by the reaction of hexamethylenediamine and sebacic acid. In this demonstration the acid chloride, sebacyl (or Sebacoyl) chloride, is used instead of sebacic acid. The equation is:
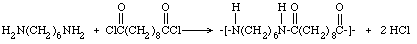
Many diamines and diacids (or diacid chlorides) can be reacted to make other condensation products that are described by the generic name "nylon." One such product is an important commercial polyamide, nylon 6-6, which can be prepared by substituting adipoyl chloride for Sebacoyl chloride in the procedure described here. The equation is:
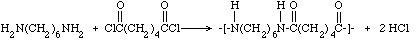
Time: About 20-30 minutes of class time.
Materials and Supplies:
- 50 ml 0.50 M hexamethylenediamine (1,6-diaminohexane), H2N(CH2)6NH2, in 0.5 M sodium hydroxide, NaOH (To prepare: dissolve 3.0 g of H2N(CH2)6NH2 plus 1.0 g NaOH in 50 ml distilled water. Hexamethylenediamine can be dispensed by placing the reagent bottle in hot water until sufficient solid has melted and can be decanted. The melting point is 39-40oC.)
- 50 ml 0.2 M Sebacoyl chloride, ClCO(CH2)8COCl, in hexane (To prepare: dissolve 1.5 ml to 2.0 ml Sebacoyl chloride in 50 ml hexane.) gloves, plastic or rubber (ones that will not dissolve in hexane)
- 250 ml beaker or crystallizing dish
- forceps
- 2 stirring rods or a small windlass
- food-coloring dye (optional)
- phenolphthalein (optional)
General Safety Guidelines:
- Hexamethylenediamine is irritating to the skin, eyes, and respiratory system.
- Sodium hydroxide is extremely caustic and can cause severe burns. Contact with the skin and eyes must be prevented.
- Sebacoyl chloride is corrosive and irritating to the skin, eyes, and respiratory system.
- Hexane is extremely flammable. Hexane vapor can irritate the respiratory tract and, in high concentrations, be narcotic.
Procedure:
- Wearing gloves, place the hexamethylenediamine solution in a 250-ml beaker or crystallizing dish.
- Slowly pour the Sebacoyl chloride solution as a second layer on top of the diamine solution, taking care to minimize agitation at the interface.
- With forceps, grasp the polymer film that forms at the interface of the two solutions and pull it carefully from the center of the beaker.
- Wind the polymer thread on a stirring rod or a small windlass.
- Wash the polymer thoroughly with water or ethanol before handling.
Food coloring dyes or phenolphthalein can be added to the lower (aqueous) phase to enhance the visibility of the liquid interface. The upper phase can also be colored with dyes such as azobenzene, but observation of the polymer film at the interface is somewhat obscured. Some of the dye will be taken up with the polymer, but can be removed by washing with water.
Disposal:
- Any remaining reactants should be mixed thoroughly to produce nylon. The solid nylon should be washed before being discarded in a solid waste container.
- Any remaining liquid should be discarded in a solvent waste container or should be neutralized with either sodium bisulfate (if basic) or sodium carbonate (if acidic) and flushed down the drain with water.