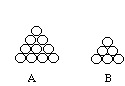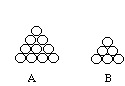Experiment 1
Crystal Packin' Mama
Crystal Structure/Packing Exercise
Objective:
The objective of this lab is to learn more about the basic crystal structures that metal atoms form.
Review of Scientific Principles:
To maximize the bonding, atoms in metals pack together as closely as possible. Several packing arrangements exist such as face centered cubic (FCC) and hexagonal closest packing (HCP).
Applications:
The properties of metals are very dependent on their crystal structure. The metal structure can be altered by processing treatments to make them more useful in various applications.
Time: 50 minutes
Materials and Supplies:
26 Styrofoam balls, about 1.5" diameter
16 toothpicks(round)
Procedure:
- Each of the Styrofoam balls will represent an atom and the toothpick will represent bonds. Attach 10 of the balls together with toothpicks to form a triangle with four balls at the base. This will form the first layer of the packing model. Draw a diagram of the arrangement of the atoms in the space below.
- Attach 6 of the balls together with toothpicks to form a triangle with 3 balls at the base. This will form the second layer of the packing model. Draw a diagram of the arrangement of the atoms in the second layer in the space below.
- Form another triangle of Styrofoam balls like the one in procedure 1 with the remaining 10 balls.
- Place the second layer on top of the first one with "atoms" of the second layer nesting in the hollows between the "atoms" of the first layer. This creates the closest possible packing of atoms.
- The third layer can be placed on top of the second layer in one of two positions. It can be placed so that its "atoms" are directly over those in layer one. This gives the ABABAB arrangement which corresponds to hexagonal closest packing (HCP). The third layer can also be placed on top of the second layer so that its "atoms" are not directly over those in the first layer. This gives the ABCABC arrangement which corresponds to face centered cubic (FCC). Try both arrangements with your layers.
Questions:
1. Which packing arrangement, FCC or HCP, is more dense?
2. What is the difference in FCC and HCP arrangements?
3. About how small would an atom have to be to fit in an interstitial hole in an FCC or HCP crystal structure?
Teacher Notes:
- It would be beneficial for the teacher to have a completed model constructed with the layers painted different colors to help the students visualize the two types of packing arrangements.
- To paint the Styrofoam balls use water based paint diluted slightly and add a small amount of detergent.
- Below are the arrangements for the triangles the students are to construct.
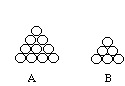
Answers to Questions:
1. Actually, FCC and HCP packing arrangements have the same atomic density. They each have approximately 26% empty space.
2. FCC has an ABC arrangement while HCP is ABA.
3. Depending on the type of hole, an interstitial atom should be approximately one third the size of the atom which makes up the crystal structure in order to "fit" well.