Basic Energy Principles
Energy is the driving force for the universe. Energy is a quantitative property of a system which may be kinetic, potential, or other in form. There are many different forms of energy. One form of energy can be transferred to another form. The laws of thermodynamics govern how and why energy is transferred. Before the different types of energy resources and their uses are discussed, it is important to understand a little about the basic laws of energy.
The Three Laws of Thermodynamics
There are three laws of thermodynamics. The first law of thermodynamics, also called conservation of energy, states that the total amount of energy in the universe is constant. This means that all of the energy has to end up somewhere, either in the original form or in a different from. We can use this knowledge to determine the amount of energy in a system, the amount lost as waste heat, and the efficiency of the system.
The second law of thermodynamics states that the disorder in the universe always increases. After cleaning your room, it always has a tendency to become messy again. This is a result of the second law. As the disorder in the universe increases, the energy is transformed into less usable forms. Thus, the efficiency of any process will always be less than 100%.
The third law of thermodynamics tells us that all molecular movement stops at a temperature we call absolute zero, or 0 Kelvin (-273oC). Since temperature is a measure of molecular movement, there can be no temperature lower than absolute zero. At this temperature, a perfect crystal has no disorder.
When put together, these laws state that a concentrated energy supply must be used to accomplish useful work.
Work
Many of us commonly think of energy as the ability of a system to do work. Work is a force applied to an object over a certain distance, such as pulling or pushing a wooden block across your desk. Your muscles do work when they facilitate body movement. Units of work and energy are joules (J). One joule equals one Newton meter (N*m).
By definition, work is an energy requiring process. So, how do you describe energy? Energy is not a substance that can be held, seen, or felt as a separate entity. We cannot create new energy that is not already present in the universe. We can only take different types materials in which energy is stored, change their state, and harness the energy that escapes from the system in order to use it to do work for us. If the released energy is not used, it will escape and be "wasted" usually as heat.
Heat
Heat is the quantity of energy stored or transferred by thermal vibrations of molecules. At absolute zero, a system has no heat energy. Heat is additive. If two masses with heat energies of 5 joules and 10 joules are added together, the added masses will have a total heat energy of 15 joules. Heat and temperature should not be confused.
Temperature
The temperature of a system is the average vibrational energy of all the molecules within the system. Temperature is not additive. Putting two metal blocks that are 75o C together will leave the new system at the same temperature. Putting two masses that are 50o C and 100o C will make the new system somewhere between 50o C and 100o C. The temperature of which would be dependent on the masses and heat capacities of each added element.
When a fast-moving molecule collides with other molecules, it loses some of its kinetic energy to those surrounding molecules. Those molecules now have more energy than they had before. This extra energy is manifested as vibrations within the molecule. Thus, the temperature of the substance being hit will increase.
Energy Conversion
Consider the explosion of gasoline in your car. The spark ignites the gas, causing combustion. Combustion of gas is the rearrangement of the carbon and hydrogen atoms in gasoline and oxygen in air into more stable forms, carbon dioxide and water vapor. The energy left over from forming CO2 and H2O propel these molecules to move faster, causing the gas to expand. The expansion of the gas causes the movement of the pistons in your car engine, which turns the crank shaft, which turns the wheels. The fast-moving gas molecules collide with the wall of the cylinder and transfer their energy to it. This energy makes the metal atoms of the cylinder vibrate faster or in other words heat up. The engine walls must be cooled or the engine will melt. Oil and water from the radiator cool the walls of the cylinder. Air from the fan cools the water in the radiator which is released into the environment as wasted energy. This wasted energy causes the efficiency to be much less than 100%.
Efficiency
Energy efficiency is the amount of useful energy extracted from a system divided by the total energy put into a system. It may also be thought of as the efficiency with which we are capable of utilizing a resource. If we don't use the energy released from the chemical bonds in a resource, the energy goes into waste heat, sound, thermal vibrations, or light. The more energy conversion steps there are in a process, the more energy you lose as waste heat. For example, in order to run your car, the chemical potential energy in the gas must first be converted into thermal energy (or heat energy) by igniting the fuel. The thermal energy is converted to mechanical energy to make the engine run. This three step process has an overall maximum efficiency of about 30%. That means that 70% of the energy initially stored in the gasoline was lost as waste heat, mostly in the form of thermal vibrations to the surrounding materials. This illustrates the importance of learning about energy and trying to find better ways to responsibly use the resources available to us.
Measuring Energy
To determine the efficiency of a process, a way must be used to measure energy. You cannot pick up energy, turn it around in your hands to describe it, or put it under your pillow to see how long it'll stay there. We do not use mechanical measurements (like how much of a certain resource is needed to make your car go so many miles at such speed) because different pathways and different machines have very different efficiencies. If we tried to quantify it mechanically, we may never know just how much absolute energy is in the resource itself. Therefore, we use the "heating value" of fuels: how using so much of a certain resource (rearranging its bonds into a more stable state) converts to so much heat (motion of molecules).
We all hear every day about counting calories. What is a calorie? A calorie (cal) is defined as the amount of heat needed to raise one gram of water 1o C. A food calorie actually consists of one kilocalorie, or 1,000 calories. Why do we worry about calories in relation to our weight? Energy conservation! If you feed your body more calories than it can use, it will store the energy in a stable state like body fat for you to use and lose later.
Energy is measured in other units as well. A common one is the British Thermal Unit, or BTU. One BTU is the amount of energy required to raise one pound of water 1o F. One gallon of gasoline contains about 125,000 BTU. A related unit is the THERM, or 100,000 BTU. Another familiar unit to physicists is the joule (J), equivalent to 0.239 calories or 9.47 x 10-4 BTU. Most systems of measurement throughout the world use joules to measure energy, even in food. When we speak in terms of energy, we often use the unit of Quads, which equals 1015 BTU. Another way energy content is often quantified is by converting the amount of energy of different sources to the amount in one barrel (42 gallons) of crude oil. Because the values are usually quite large, the equivalence is usually compared to so many million barrels of oil per day (MBPD). Burning 500 million tons of coal a year would be approximately 6 MBPD of oil for a year. Currently the United States is using about 18 MBPD, or 6.5 billion barrels per year!
Table 2: The average energy contained in or consumed by some common items. * Note that 1015 BTU = 1 QUAD
Average Energy In Btu Of...
| A Match | 1 |
|---|---|
| An Apple | 400 |
| Making A Cup Of Tea | 500 |
| A Stick Of Dynamite | 2,000 |
| A Loaf Of Bread | 5,100 |
| A Pound Of Wood | 6,000 |
| 100 Hours Of Television | 28,000 |
| A Gallon Of Gasoline | 125,000 |
| 20 Days Gas Cooking Range | 1,000,000 |
| Food For 1 Person For Year | 3,500,000 |
| Heat St. Louis House For Year | 90,000,000 |
| Apollo 17 To The Moon | 5,600,000,000 |
| Hiroshima Atomic Bomb | 80,000,000,000 |
| 1000 Transatlantic Jet Flights | 250,000,000,000 |
| 1 Year Oklahoma Energy | 1,000,000,000,000,000 * |
| 1 Year Energy 30 African Countries | 1,000,000,000,000,000 |
| Energy Used By U.S.1993 | 83,960,000,000,000,000 |
| Energy Used By World 1993 | 343,000,000,000,000,000 |
Forms of Energy
Energy exist in many forms. Table 1 gives different types of energy along with their definitions.
Table 1: Forms of Energy
| Energy form | Definition |
|---|---|
| Chemical Energy | Energy stored in chemical bonds of molecules. |
| Thermal or Heat Energy | Energy associated with the heat of an object. |
| Mechanical Energy | |
| Potential Energy | Energy stored in a system. |
| Kinetic Energy | Energy that is from motion of matter. |
| Electrical Energy | Energy associated with the movement of electrons. |
| Radiant or Solar Energy | Energy that is from the sun. |
| Nuclear Energy | Energy found in the nuclear structure of atoms |
Chemical Energy Basics
When we use a resource, such as coal, to produce energy, we are breaking the chemical bonds within the substance and rearranging them into more stable bonds. This change results in the formation of different products, such as carbon dioxide and water in the case of combustion, and a release of energy.
That may sound complex, but this analogy makes it really simple. Picture an old-fashioned water well. The molecule is at the bottom of the well. It takes energy to bring it to the top of the well (winding up the bucket). Think of the molecule as now being broken up into its atoms--the energy that was expended to do that is its binding energy, or the energy holding the atoms together in a molecule. One way to measure binding energy is the heat of formation. Now those independent atoms (at the top of the well) combine into other molecules that are even more stable. Combining means that they fall back down into a couple of new wells. These wells are deeper than the original well--there is more binding energy in these new molecules. When the atoms "fall down" into the new wells, becoming new molecules, energy is released. The hand-crank spins wildly as the bucket falls to the bottom. To figure out how much net energy is released, just compare the depths of the new wells to the old one.
A numerical example may help explain this. The combustion of methane to carbon dioxide and water is represented by the following chemical reaction:
CH4 + 2 O2 + spark --> CO2 + 2H2O + heat
The heat of formation of CH4 is -17.88 kcal/mol. The heat of formation of O2 is defined as 0 kcal/mol. The sum of the heats of formation of the reactants (-17.88 kcal/mol + 0 kcal/mol) is the sum of the depths of the original wells in the previous example. Adding a spark to the left side of the reaction is analogous to cranking the bucket to the top of the well and expending energy.
Carbon, hydrogen, and oxygen atoms are now at the top of the well. They then combine into other molecules, namely CO2 and H2O. In combining, the molecules fall into new "wells" whose depths correspond to the heats of formation of the new molecules. The well for CO2 has -94.1 kcal/mol, and the well for H20 has 2(-57.8 kcal/mol) because there are two moles of water formed for each mole of methane burned. The combined depth of these two new wells for the products is -209.7 kcal/mol, which is deeper than the wells the molecules came from.
With numbers, the equation above looks like:
-17.88 kcal/mol + 0 kcal/mol = -94.1 + 2(-57.8) kcal/mol + heat
Now rearrange:
heat = -17.88 kcal/mol + 94.1 kcal/mol + 2(57.8) kcal/mol
= +191.82 kcal/mol methane burned
This means that there is 191.28 kcal per mole that is now expressed as heat and the motion of the products, CO2 and H2O.
Fossil Fuels
Fossil fuels are coal, oil, and natural gas. We call them fossil fuels because all of them in some way originated from the decomposition of organic matter in or on the earth. Each provides a unique source of energy that humans have taken advantage of over thousands of years. Approximately 90% of our energy consumption comes from fossil fuels. Approximately 50% of the fossil fuels humans have consumed throughout history were used in the last 20 years. The scary fact is that we cannot make more. In other words, our fossil fuel reserves are finite. Humans have used most of our reserves in a 200 year period. We will need to convert our energy usage to the most plentiful fossil or non-fossil fuel sources over the next hundred years in order to meet the world's growing energy needs.

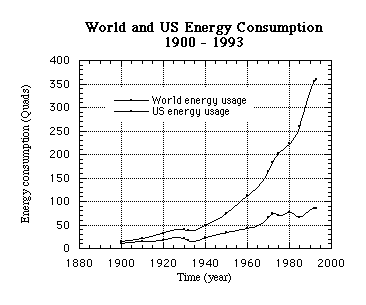
Figure 1: World and US Energy Consumption (1900-1993) represents the approximate amount of energy in Quads used by the world (top line) and the United States (bottom line).
As you can see, the energy consumption of the world has taken a dramatic increase since 1900. The dip in the curve in the 1930's represents a decrease in energy use due to the Great Depression. In 1973 the United States also experienced decreases in usage due to increased oil prices as a result of the Arab Oil Embargo and again in 1983 with increased oil prices. The world energy consumption was not affected as greatly and is increasing at a greater rate because now many third world countries are increasing their energy usage as they industrialize. The United States' consumption is more steady because most of our industrial development has already occurred.
Coal
Coal is composed primarily of carbon. It is formed from dead plant matter which decomposes into peat in swamps over millions of years. The peat is continually buried under other matter, mud, and sand in non-marine environments. This burial causes it to decompose as its bonds recombine and rearrange. With increases in pressure and temperature, coal seams form. Several different types of coal can be found depending on the depth and location of the seam. The four main types of coal, which differ by carbon content are shown in the following table.
Table 3: Types of Coal by Sulfur Content (Hinrichs p458)
| Type of Coal | Carbon % | Energy Content (Btu/ lb) |
|---|---|---|
| Lignite | 30 | 5000-7500 |
| Subbituminous | 40 | 8000-10,000 |
| Bituminous | 50-70 | 11,000 - 15,000 |
| Anthracite | 90 | 14,000 |
Lignites are the "youngest" coals, which have high water content and low heating values. The heating value of a fuel is used to quantify the useful energy content of different fuels. Lignite often has many impurities and is therefore not a preferable type to use. Subbituminous coal is cheaper to mine because it is not as deep as bituminous coal and contains less sulfur than lignites. Bituminous coal is the most abundant type of coal. It has a high heating value, but it also has a high sulfur content. Anthracite coal is a very hard coal which burns longer, with more heat and with less dust and soot than the other types of coal. These qualities make anthracite a popular home heating fuel.
Not all plant material turns into coal; some eventually becomes graphite, and a tiny amount is compressed into diamonds. Coal is burned in power plants to produce heat which is used to change water into steam. This steam turns a large fan-like structure called a turbine which generates electricity.
Coal Reserves
Coal reserves are located all over the world. Although the United States has approximately 22% of the world's coal reserves, coal only supplies about 24% of the our energy needs. Coal usage worldwide is increasing due to high oil prices and skyrocketing demand for oil in the transportation sector. Electrical utilities consume about 87% of the total coal produced, and about 55% of electrical power generated from fossil fuels comes from coal. Coal resources are projected to last from between 400-450 years. Coal is measured in short tons, 2000 lbs each, which are equivalent to approximately 3.5 barrels of oil. One short ton can provide approximately 26 x 106 BTU.
Mining
Coal is commonly recovered from the earth by two methods. Surface mining, or strip mining, is preferred due to cost and safety factors. Strip mining usually occurs on flat land. Hilly or steep terrain requires contour mining. Deep mining involves digging shafts and tunnels to gain access to the coal seam. The coal can then be excavated from the seam with only some columns of earth left behind for support. Deep mining is unappealing because of safety hazards in the tunnels and health hazards like black lung disease. However, deep mining has become more acceptable due to automation. On the flip side, the negative effects of mining are the intense, irreversible damages inflicted upon the environment.
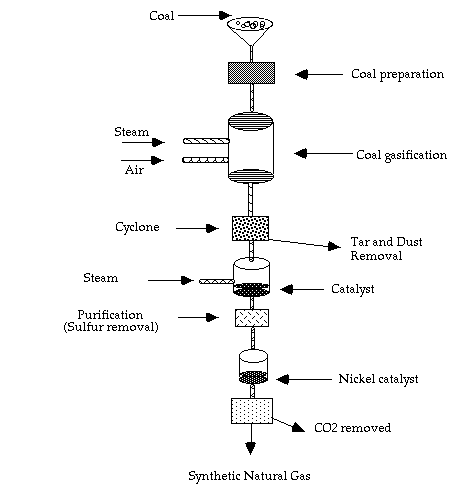
Coal Gasification
Coal gasification is a process by which coal is converted into a synthetic fuel, natural gas. The process basically adds hydrogen to the carbon in coal. In order to change the carbon to hydrogen atomic ratio from 12 to 1 in coal to 1 to 4 in natural gas, several steps must be carried out. First the coal is brought into contact with high-pressure, high-temperature steam in the gasifier. The heat for the reaction from coal to "synthesis gas" is provided by introducing some oxygen, which causes some of the coal to burn. In the second stage, the C:H ratio is increased by further addition of steam, which increases the heating value of the fuel. The resulting mixture is then purified and converted to methane in the presence of a nickel catalyst. The methanization is an exothermic reaction, in which lots of low temperature heat is lost, therefore making the process inefficient. Synthetic methane is the resulting fuel. See Figure 2 for a schematic of a coal gasifier.
Coal Liquefaction
Coal liquefaction converts coal into synthetic crude oil, or syncrude. This process also involves adding hydrogen to heated coal and then separating the gas and liquid product. The hydrogen is added to coal in a slurry at elevated temperatures and pressures. The high temperature breaks the carbon bonds, which produces a liquid phase product due to the high pressure.
Economical considerations hinder the further development of coal gasification and liquefaction systems. It has been neither economical nor efficient to produce synthetic fuels from coal on a large scale basis. The production facilities are more expensive to run and maintain than simply buying the oil or natural gas itself. It is important, however, that the processes are maintained and improved even at a slow rate. As our reserves of crude oil are depleted, the price of oil will probably increase dramatically, making the use of synthetic fuels more economical.
Oil
Oil is mainly a mixture of hydrocarbons and is formed by the deposition of dead plant, animals, and marine microorganism matter in or near marine sedentary basins. Once the matter is buried at about 450 meters, the temperature and pressure begin to cause the rearrangement of matter. The newly-formed liquid molecules migrate through porous rock formations such as limestone and sandstone until they are trapped by a non-porous rock barrier. Crude oil is a mixture of hydrocarbons with some oxygen, nitrogen, and sulfur impurities. Sulfur content of crude varies and is either designated "sweet" (less than 1% sulfur) or "sour" (higher than 1% sulfur). One barrel of oil is equivalent to 42 U.S. gallons and can provide about 6 million BTU, or about 143,000 BTU per gallon.
Reserves
Crude oil reserves are located all over the world, but the Middle East alone has about 63% of the known reserves. Saudi Arabia contains by far the most oil with the equivalent of 1,545 Quads of proven resources available. Even though the United States has only about 161 Quads of proven resources left, we have more wells than any other country in the world. Our use accounts for about 25% of the world's oil consumption, while we only have 3% of the world's oil. We import around 40% of the oil we use. In 1994, 65% of oil used in the United States was used in transportation. Only 3% was used for the generation of electricity, with the other 32% going to industrial, commercial, and residential uses. We rely so heavily upon oil products for transportation purposes because it is an easily transportable liquid, we already have an efficient distribution system set up, and there is an entire fleet of vehicles on the road that use gasoline for operation.
Recovery
Petroleum is usually recovered by drilling wells down through the non-porous rock barrier under which the oil is trapped. There are three types of oil recovery. Primary oil recovery occurs as the oil flows out of a well by its own pressure or is pumped out. This removes about 30% of the oil. Another 10% is removed by flooding the well with high pressure water or gas, a method of secondary recovery. Some methods of tertiary recovery have been developed in which the oil is heated (by burning some underground detergents or the oil itself) to scrub it out. This only removes another 10%, however, and requires energy to do so. Therefore, about half of the oil is left trapped in this rock with no economical means for its recovery.
Unconventional oil recovery entails obtaining oil from oil shale. Oil shale is a material with hydrogen content between that of coal and crude oil due to the fact that it was never buried deeply enough or heated enough to form crude oil. The concentration of oil in this material is quite low, and it is chemically bonded to the shale. The maximum amount of recoverable oil is one barrel per 2.4 tons of sand or 1.5 tons of rock. Enormous problems also occur with extraction of oil from oil shale. The advantage is that 20% of the United States contains oil shale. The potential amount of oil contained in oil shale is greater than the known and unproved crude oil resources in the world, which would add approximately 40 years to the projected time before oil will be exhausted. The dilemma is that it takes about half the energy contained in the shale to extract the oil. Prices for barrels of oil from oil shale can range from $40 to $80 per barrel, whereas normal crude oil cost about $18.75 per barrel in June 1995. Recovery of oil from oil shale is therefore not economically feasible at this time.
Refining
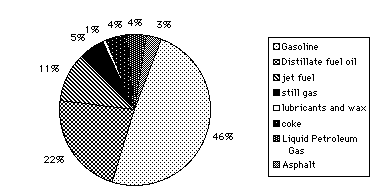
Crude petroleum is refined by fractional distillation. This means that the distillation process occurs with successive separations carried out at increasingly higher temperatures. The condensed vapors are collected in several portions, or fractions, the first fractions being richest in low boiling point components. These include gasoline, kerosene, furnace oil, naphthas (liquid hydrocarbon mixtures), and lubricating oils. The heavy residues left over are used as asphalt and residual oil. The second step in oil refining is conversion, or cracking, of the molecules in order to squeeze out a higher percentage of lighter, low boiling point products like gasoline from each barrel of oil. The last step, treatment, or enhancement, increases the quality of the product by such means as removing sulfur from kerosene, gasoline, and heating oils. The distribution of products is shown in the following graph, Figure 3.
The crude petroleum that we recover is refined into useful products for the production of electricity as well as for use in machinery and equipment, such as automobiles. Many products we use every day are produced from petroleum such as ink, crayons, bubble gum, dish washing liquids, deodorant, eyeglasses, records, tires, heart valves, and more!

Oil in the Twentieth Century
We rely heavily on oil, and it has fueled most of the global energy consumption since World War II. Past trends have been governed by both economical and political occurrences. The Great Depression marked a decrease in the total amount of fuel used in the United States, and oil consumption decreased accordingly. Nevertheless, the U.S. demand for oil has increased greatly with our energy needs. In 1950, oil accounted for less than one third of the world's energy use, but today it accounts for almost half. The last two decades have been truly pivotal in world oil consumption and prices. In 1960, a cartel called the Organization of Petroleum Exporting Countries, or OPEC, was formed by the oil-producing states of Algeria, Ecuador, Gabon, Indonesia, Iran, Iraq, Kuwait, Libya, Nigeria, Qatar, Saudi Arabia, Venezuela, and the United Arab Emirates. OPEC's influence on petroleum prices steadily grew until, in the 1970's, OPEC was able to set their own prices for their exports and take the control of oil away from foreign companies.
Several events marked the dramatic increase of oil prices, which still remain in effect, long after the political situations were resolved. The Arab-Israeli war prompted the Arab Oil Embargo of 1973, during which the Arab OPEC countries cut back on production and refused to export their oil to several Western countries including the United States. This caused the price of oil per barrel to triple from about $8 to $25. Then in 1978 and 1979, the Iranian revolution caused a disruption in production, and prices doubled from $22 to $44 per bbl. These events are reflected in the World and US Energy Consumption chart (Figure 1, p 9). In response, the countries of the world placed a greater emphasis on reduction and conservation of energy use. Thus, the world's dependence on OPEC fell, and the prices began to decline around 1981, continuing until the Iraqi invasion of Kuwait in 1990, which prompted a sudden increase in the price of oil on the world market. Prices then began to fall again as other countries increased production to make up for the drop in production in Kuwait. In June 1995, the cost of oil was approximately $18.75 per barrel.
The low cost of oil and its adaptability to many uses make it a key fuel for expanding economies. The United States, however, depends less today on oil for its fuel mix, and more on coal, natural gas, nuclear, and renewable technologies. Our transportation industry still relies heavily on oil as its chief source of energy. In order to significantly decrease usage, the American public will need to be encouraged to depend less on their cars and more on public transportation. Especially in big cities, car pooling to work and public transportation can reduce the amount of oil consumed.
Natural Gas
Natural gas is 99% methane (CH4) and 1% other light hydrocarbons such as ethane, (C2H6), propane, (C3H8), and butane, (C4H10), as well as some aromatic hydrocarbons. Natural gas is the gaseous component of coal and oil formation. Uses of natural gas range from industrial and commercial heating and power to residential heating, cooling, and making the blue flame in our kitchen burners (gas stoves). Places in the earth where large deposits are found are called reservoirs.
Reserves
Natural gas is either found mixed in oil or it is released from coal. It is measured in cubic feet, ft3. The energy content in 6,000 ft3 of natural gas is the equivalent of one barrel of oil. World reserves are greatest in Russia, followed by Iran, and the United States. The Annual Energy Outlook in 1994 estimates that world gas reserves are over 30 trillion cubic feet, approximately equivalent to 5,000 Quads of energy. Eurasia and the Middle East have about three-fourths of the world's natural gas. The rest is fairly evenly distributed among other regions of the world such as North America, Central and South America, Western Europe, Africa, and Asia/Pacific. Known reserves are projected to enlarge as previously unexplored regions are explored. Projections for the longevity of natural gas range from 80 to 135 years. Remember that every projection of how long any fossil fuel will last is dependent on the actual growth rate of consuming countries and the trends of consumption and economics of the other fuel sources.
Recovery
Wells for natural gas are drilled in underground reservoirs of porous rock. When it is removed from a reservoir, natural gas can either be pumped to the processing station for removal of liquid hydrocarbons, sulfur, carbon dioxide, and other components, or stored in large caverns underground until it is needed. Pipelines are the principal method of natural gas transportation. Because natural gas was not highly valued in the early half of this century, the expansion and development of the pipeline system did not occur until about 1940. In addition, natural gas deposits were often burned off when they were found mixed in oil deposits. Today gas pipelines cover the United States, Canada, Western Europe, and Russia. Transporting it overseas has been made more economically expedient by liquefaction techniques, but it is still quite expensive. The price of oil has been a key factor in determining the development of transportation facilities for natural gas.
Environmental Concerns
Many environmental problems are associated with burning fossil fuels for energy. The combustion reaction of a fossil fuel with oxygen releases water, carbon dioxide, and any impurities contained in the resource into the environment. This section discusses some of the major environmental concerns arising from fossil fuel use.
Coal
Although coal is abundant, its impurities pose potential dangers to the environment, in the form of acid rain, destruction of topsoil fertility and/or global warming. Since the 1960's, increasing importance has been placed on the need for a cleaner use of coal for environmental reasons.
Acid Rain
The high sulfur content in coal is primarily responsible for acid rain. When sulfur-containing coal is burned, the sulfur is usually emitted as sulfur dioxide, SO2 (1).
The SO2 then reacts with the air, becoming oxidized to sulfur trioxide SO3 (2) and then reacts with water to form droplets of sulfuric acid, H2SO4 (3).
S + O2 --> SO2(1)
2SO2 + O2 --> 2SO3(2)
SO3 + H2O --> H2SO4(3)
The sulfuric acid is absorbed by fine particulates in the air along with water vapor. The fine particulates, often formed from the ash of burnt coal, provide an excellent surface for catalytic reactions to occur, producing more H2SO4. The acid then falls as acid rain or even as dry deposition, known as acid dust.
Acid rain has negative repercussions on both humans and the environment. Increasing acidity of lakes may cause the death of aquatic plants and animals. Acid rain also damages vegetation in four ways:
Man's health is also directly affected by acid rain, manifested by afflictions from a minor skin irritation, to severe lung damage, and possibly death. Acid rain also affects our lives in less serious ways. Most people who rollerblade know how annoying it can be to skate on a sidewalk with pits in the surface. Those pits are largely the result of acid rain. The problem of acid rain is not limited to the United States. In Europe many of the marble monuments of the Greek and Roman empires are being destroyed by the effects of acid rain. When fossil fuels with nitrogenous impurities (NOx) are burned, the nitrogen reacts with water to form nitric acid. This is a particular problem in urban centers where many automobiles that emit oxides of nitrogen are operated daily.
In an attempt to regulate the amount of sulfur and nitrogenous compounds in the air, the government has placed many regulations on power plants in the form of Clean Air Acts. As a result, the amount of acid rain present has decreased as well. What have coal power plants done to reduce the sulfur emissions? Many plants use a combination of methods to decrease emissions of sulfur and nitrogenous compounds:
Global Warming
In addition to acid rain, coal power plants pose yet another problem to the environment known as the greenhouse effect. The combustion of coal produces three primary products: ash, H20, and CO2. In the atmosphere, these gases trap infrared radiation from the earth which would, under normal circumstances, radiate out to space. The consequence of greenhouse gas build-up is global warming, which may adversely affect the earth's climate. The increasing overall temperature could possibly cause warming in the oceans or melting of polar ice caps. Plant and animal life could be greatly affected by these changes. Although estimates vary, experts project a possible 2o C increase in the earth's temperature by the year 2050. This rate of increase is approximately 7 times the normal historical rate of about 0.04o C per decade over the last several hundred years. The question at hand is how much and how fast the temperatures will rise. Destruction of rain forests contributes to this effect because trees are an excellent sink, or utilizer, of CO2, as it is a requirement of photosynthesis.
Unfortunately, coal power plant scrubbers cannot take care of CO2 emissions. Planting more trees would be an obvious way to create users of CO2. However, because carbon dioxide is a natural product of combustion of a fossil fuel with oxygen, the only real way to reduce the amount of carbon dioxide output is to reduce fossil fuel consumption.
Oil and Natural Gas
As with any fossil fuel, environmental considerations such as pollution and harmful emissions arise with the use of petroleum products. Smog is primarily created by hydrocarbons along with carbon monoxide (CO) and other complex toxic molecules from oil processing, all of which are emitted from the exhaust pipes of cars. As the problem with emissions has increased with the growth of automobile use, government regulations of the acceptable amount of emissions have multiplied accordingly. Auto engineers have endeavored to design cars which burn gasoline more cleanly and efficiently and incorporate more advanced filter systems. Car manufacturers have developed automobiles which are capable of reducing hydrocarbon emission by 97 to 99%. They are also making strides to develop "pollution-free" cars, which have modified pollution control devices. Manufacturers are also experimenting with cleaner burning fuels like methanol and natural gas. Other engineers have designed solar and electric cars. However, each fuel source presents a unique set of problems.
Worldwide crude oil distribution and trade occurs mainly by sea. This presents a potential problem in the form of oil spills in the oceans, threatening coastal and marine plants and wildlife in addition to coral reef degradation by anchoring tankers.
In the past decade there has been a movement to expand natural gas usage for several reasons. There are greater domestic reserves of natural gas, and it is the cleanest-burning fossil fuel because it contains the least amount of carbon and virtually no sulfur. Since natural gas is a clean burning fuel, some new cars are designed to use it. Natural gas costs about two-thirds as much as gasoline per BTU, and it is in comparatively plentiful supply. However, in order to convert to natural gas use for transportation, fuel tanks would have to be pressurized, making them heavier and larger. Cars would also need to be refueled about every 100 miles because natural gas has a lower energy density than gasoline.
Equipment and technology are being developed for natural gas use in the energy -demanding sectors of electricity generation, transportation, and residential and commercial cooling. Electrical generation by natural gas has been enhanced with the development of combined-cycle systems in which a natural gas-fueled combustion turbine combined with a heat recovery steam generator and steam turbine work to efficiently produce electricity in two ways. These systems produce less than half the CO2 per kilowatt hour than do state-of-the-art coal power plants.
Renewable Energy Sources
A renewable resource is a fuel source that can provide energy for man forever if man takes care of it. There are many types of renewable resources that man has learned to take advantage of, ranging from solar power to biomass and geothermal to wind power. With these resources, man has utilized the power of the sun, wind, and the earth itself. The efficiency varies with the resource, however.
Passive Solar
One quite common application of renewable energy is passive solar heating for the home. A passive solar heating system collects energy from the sun and uses this energy to heat a space directly, such as a sun room, or to heat a fluid. If a fluid is heated, it is considered a source of thermal mass for the system, which can be used later to radiate the heat to the surroundings. The five main components of passive solar heating are the collector, absorber, storage mass, distribution system, and control system. In the case of a sunroom, the collector is the double layer of glass in thermal window panes that trap the heat in and reduce the amount of heat loss by convection. The absorber consists of several parts: surfaces of the walls, floors, and any water-filled containers inside the sunroom. The storage mass, or thermal mass, is the building material such as concrete, brick, and any water. Thermal mass usually has a high heat capacity to retain the heat absorbed during the daylight hours, and radiate it at night. One method of increasing the thermal mass of a house is to build the house partially underground. A distribution system of fans or vents should be installed to move the heat by natural convection. Control systems, or heat regulation devices such as movable insulation, roof overhangs, thermostatically controlled fans, and adjustable vents are used to control the temperature of specific rooms. Hot water heating systems may heat a primary fluid that will not freeze in the winter, and pump that fluid to a heat exchanger to heat water for use inside a building. Many innovative passive solar heating systems have been designed to optimize the efficiency of the passive solar home.
Active Solar
Another method of capturing the sun's energy is through the use of active solar systems, or photovoltaic systems. Photovoltaic systems use solar cells to directly produce electricity from solar radiation. The photovoltaic cell is made by using two semi-conducting materials which have different types of charge carriers, so when they are placed back-to-back a potential difference can be measured. The boundary between the two conductors is called a depletion region. If a photon, or "bundle" of electromagnetic energy, from the sun strikes an electron near the boundary between these elements, that electron moves to a higher energy level. The potential difference moves this free electron leaving an unoccupied space, or "hole." The entire process causes a charge separation. As the electron and the hole move across the photovoltaic cell and travel through the wire connected to what's being powered (a solar calculator for example), an electrical current in the circuit is produced.
This system is very cost effective in situations where electric power is not readily available and the power requirements are relatively low. It is roughly estimated that about 10,000 square miles of solar cells could produce enough energy for the entire nation! So, why haven't we converted our energy usage to active solar? 10,000 square miles of solar cells is a lot of solar cells! They would have to be built, a maintenance system would be required in order to maximize efficiency, the weather would have to be dependable, and it would be expensive. It has not been economical to develop this source because the cost of the cells is too high. Perhaps as the replacement of fossil fuels becomes essential, this technology may be better developed. Many homes and businesses use solar cells to reduce their electric bill. Solar cars have also been developed as a result of competitive races between major universities.

Figure 4: A solar-powered calculator is an example of a complete active solar system. When a photon strikes the cell, an electron is excited and becomes free to move, leaving a "hole." This causes one electron after another to fill the hole. The electron moves one way and the "hole," or lack of electron, moves the other way. This creates a current that is directed through the wire to the calculator. The circuit is now complete, and your calculator turns on.
Solar Ponds
Solar ponds are also utilized to capture the sun's power. A solar pond uses the principles of energy transfer by convection to heat water to steam for heat production. The bottom of the pond is dark colored in order to absorb the sun's rays. The pond is filled with saline water made with NaCl, MgCl2, sodium carbonate, or sodium sulfate. A gradient is maintained at varying densities. The bottom is the most dense and is used as a storage zone. It is convective and can store a working temperature of up to 80-85o C. Above the bottom layer is a nonconvective zone, or insulation zone, with a density gradient which facilitates a temperature gradient as well. This layer functions as insulation. There is no convection in the gradient layer because even though the warm water would normally rise, the high salt concentration at lower levels does not allow the water to be light enough to float up as it warms. This prevents heat in the bottom from reaching the top of the pond. The top layer, or surface zone, is convective due to wind-induced mixing and daily heating and cooling. These layers are represented in Figure 5 on the following page. The hot brine, or salt water, on the bottom may be extracted and used for direct heating and low-temperature industrial uses like drying crops and agricultural shelter heating. The problem with solar ponds is that it is essential to have a controlled saline density gradient, which is quite difficult to maintain. Additionally, the pond must be kept free of dirt and other light-absorbing materials. Thus, for large scale operations, the difficulties are too great to rely upon solar ponds for efficient heat production.
Here is one possible system for converting the heat energy from the salt water to electricity. An organic fluid is heated and boiled as it is pumped through tubes in an evaporator. The hot brine is pumped from the bottom of the solar pond through the evaporator (where it transfers heat to the organic fluid), and returned to the pond. The organic fluid, which is now a vapor, has sufficient pressure to spin the turbine and generator. The vapor has transferred some of its kinetic energy to the turbine. The cooler vapor is pumped to the condenser where it is condensed to a liquid as it transfers energy to the cold water being pumped through the tubes of the condenser. The organic liquid is now pumped to the evaporator to continue the process. As the gradient layer diffuses as time passes, new freshwater and salt water can be pumped into the pond to maintain a sufficient gradient layer.

Wind Power
Not only does the sun supply the earth with direct radiant energy, but it also heats the gases of the atmosphere, which produces wind. Wind power uses energy from the moving air to turn large blades on windmills. In the past, the motion of the blades was used to grind flour or pump water, but now the blades turn turbines, which rotate generators in order to produce electricity. Wide open windy spaces are needed in order for this system to be efficient. Wind energy produces no air or water pollution, involves no toxic or hazardous substances, and poses no threat to public safety. The major problem with wind power is the limited ability of sites with steady wind and the lifetime of the wind power generator units.
Hydropower
Flowing water can also be used to generate power. Hydropower systems use the energy in flowing water for mechanical purposes or to produce electricity. The head and the flow are two variables that essentially determine the potential efficiency of a site for a hydropower system. The height of the falling water is called the head. The greater the head, the higher the velocity of the water falling will be, and hence the greater the pressure with which it hits. The flow is the total amount of water moving. The advantages of using water for a power source are that the resource is free, and it can be stored effectively and put to use quickly. Water for hydropower may be stored in a reservoir or above a dam forming a lake. The mechanics of the hydropower system are very similar to the wind power system, the only difference being water turning the blades instead of wind. The turning wheel can also drive a shaft to produce mechanical energy, which may be used to perform simple tasks, especially for agricultural applications such as sawing wood or grinding grain.
Most sites available for large scale hydroelectric plants have already been developed. Therefore, new developments in hydroelectric generation are small scale. Small scale hydropower systems are quite efficient when used to supply local needs. The Hoover Dam is an example of a large scale hydroelectric plant still in use in the United States today. The United States and Canada have the greatest number of hydroelectric plants. Virtually every other country in the world has some development of hydropower plants. Hydroelectricity is the power source of choice for many developing countries. However, the development of hydroelectricity is quite expensive as well as site-dependent. There are some concerns over the detrimental environmental effects of hydroelectric power. Environmental problems include siltation and erosion, the breaking up the free passage between oceans and rivers, weed growth, disease spread by small organisms that live in stagnant water, and floods due to dam failures.
Geothermal
Geothermal energy originates from the inner core of the earth. Geothermal energy is evident on the earth's surface in the forms of volcanoes, geysers, and hot springs. Even though the amount of energy within the earth is basically infinite, our ability to use it is limited by site considerations. Favorable sites for geothermal energy extraction are rare and occur where magma, or hot molten rock of the earth's mantle, has been pushed up near the earth's surface through faults and cracks in the crust. The resulting "hot spots" 2 to 3 km from the surface naturally heat water that leaks in. From there, the steam and hot water may be used directly to turn turbines or to heat homes. As the steam and hot water is expelled from the hot spot, cooler water runs back down, and the cycle continues.
Biomass
Biomass materials are the remains of animal or plant life that have not been subjected to the tremendous heat and pressure that formed the fossil fuels. Biomass materials such as wood, dried dung, animal wastes, and even garbage can be used as renewable sources of energy to heat homes, cook food, and even produce electricity. The energy produced when wood is burned was originally stored in the bonds of the glucose formed during photosynthesis in the leaves of the tree. As the wood is burned, energy is required to break the bonds in the cellulose. As the carbon dioxide and water are formed, more energy is released than was initially required to break the bonds. Thus, net energy is released. (Recall the "well" analogy.) Examples of widely-used biomass energy systems are ethanol in gasoline, anaerobic digestion of municipal waste water or swine waste to produce methane gas, and incineration of garbage.
There are several other applications of biomass fuel. Ethanol is presently used in gasohol (10% ethanol, 90% gasoline), resulting in a cleaner burning fuel, which emits an average of 20% less carbon monoxide than unblended gasoline. Swine farmers collect the manure and add methane-producing bacteria called methanogens in a controlled environment. The methane is collected and burned to produce electricity for the hog confinement buildings. Municipal waste water treatment plants use a two stage process to separate the waste into two parts. One converts waste sludge into a variety of organic acids and carbon dioxide; the other reacts the waste with the methanogens to produce methane. The forest industry now burns much of its waste to provide heat and electricity.
Important Considerations
Unfortunately, use of renewable resources can be greater than its rate of renewal. For example, a biomass resource such as corn for ethanol production is dependent on the yield of corn crops. In the case of wind power, the wind must blow at a specific speed and from a specific direction for optimum efficiency. Hydropower is renewable as long as it rains. The efficiency is dependent on the depth of the water behind the dam, which is also site dependent. Further, in a reservoir-based hydropower plant, silting may occur, which would slowly decrease the potential of the power plant.
Even passive or active solar systems are dependent on the weather conditions and the number of daylight hours. For optimum efficiency, photovoltaic systems must be mounted on a movable base to keep the surface of the solar cell perpendicular to the sun's rays. In addition, the electrical energy produced in the daylight must be stored in batteries for use at night. The efficiency of passive solar systems, such as sun spaces and solar water heaters, is greatly affected by climate, season, and building orientation with respect to the sun. Solar heating systems that use fluids and heat exchangers must be designed to withstand the temperature range of the site. For example, in colder climates, antifreeze must be added to water to prevent freezing and subsequent pipe bursting. In the case of sun spaces, the room should be facing south with shade from the roof overhang or exterior structures or trees in order to prevent overheating in the summer.
As long as the sun shines, it will produce wind, rain, and biological growth. The amount of energy reaching the outer atmosphere of the earth from the sun is quite predictable and dependable. The difficulty is that the amount of usable solar energy received on a specific day at a specific site is very unpredictable and undependable. If there are cloudy skies, the solar radiation is said to be diffuse radiation which cannot be used efficiently even though it is there. Weather conditions and site conditions including natural disasters such as floods, earthquakes, and hailstorms greatly limit the dependability of solar energy. Thus, solar power plants may not be the magical alternative source to replace fossil fuels for large scale use. However, small scale use of renewable resources to supply energy needs can reduce dependence on fossil fuels.
As the reserves of fossil fuels continue to diminish, alternative energy resources are being developed. Nuclear power is one example of an alternative energy resource. However, public health and safety considerations concerning nuclear power plants are abundant. The purpose of this section is to describe the mechanics of nuclear power as well as to answer some commonly asked questions about nuclear power.
Basic Nuclear Principles
The atom is composed of subatomic particles called protons, neutrons, and electrons.

Figure 6: Schematic diagram of an atom. The protons and neutrons are located in the center, and the electrons orbit the nucleus. The electrons are located in an electron cloud surrounding the nucleus, represented by the oval orbits.
NOTE: The atom is not drawn to scale: the atom is approximately 100,000 times the size of the nucleus.
Remember that like charges repel. So how can positively charged protons exist together in a nucleus? There is a force called the strong force that holds protons and neutrons together at very small distances. The electrons are "bound" to the nucleus by electric charge because the unlike positive and negative charges of the protons and electrons, respectively, attract.
Two isotopes of an element contain the same number of protons and electrons, but a different number of neutrons in the nucleus. An isotope with more neutrons is called "heavy" in comparison with the "lighter" isotope (of the same element) with less neutrons. Take, for example 235U and 238U (read "Uranium 235" and "Uranium 238"). Both consist of 92 protons and electrons, which makes them Uranium. The difference is that 235U has 143 neutrons (235-92) whereas, 238U has 146 neutrons (238-92). Therefore, 238U is heavier than 235U.
Nuclear energy may be defined as the energy found within an atomic nucleus or as the nuclear binding energy. If we take the helium nucleus, the mass of its parts (two protons and two neutrons) is less than the mass of the nucleus itself. Using Einstein's equation E = mc2 (where c is the speed of light) we can see that a tiny bit of mass can make an enormous amount of energy.
In a nuclear reaction, an unstable nucleus will become more stable by emitting particles and rearranging the neutrons and protons into more stable nuclei. A stable atomic nucleus does not undergo nuclear reactions unless bombarded with nuclear particles such as protons, neutrons, or alpha particles, a process called nuclear bombardment.
Radioactivity Basics
Being a science student, you are undoubtedly familiar with the periodic table of the elements. When you read the atomic weight of an element on the chart, such as carbon at 12.01 g/mol, you are not reading the absolute atomic weight of the element. Instead, you are reading the average of the naturally occurring isotopes of carbon, some of which include radioactive isotopes. Remember that an isotope of an element has the same number of protons and electrons as the other isotopes, but has a different number of neutrons in the nucleus. This sometimes causes the isotope to be unstable and to radiate energy, or be radioactive. The half-life of a radioactive substance is the amount of time it takes for the substance to emit one half of its radioactivity.
Radioactivity is all around you, from the food you eat to the bricks in the buildings surrounding you. Radioactive elements that occur naturally are considered part of background radiation. Background radiation comes from anything that is part of the natural world that is around all the time. Because of this, you can easily conclude that all radioactivity is not bad. Rather, your body is bombarded with radioactivity every minute of every day, especially if you get lots of exposure to the sun. Several every day ordinary food objects are slightly radioactive, including table salt substitute and bananas. Check it out the next time you take your Geiger counter to the grocery store! Another common radioactive object sold in stores is the Coleman Lantern mantle, which contains thorium.
The "dangerous" radiation comes in eating or otherwise ingesting radioactive elements which occur in large concentrations, or from external sources which give a high dose.
Measuring Radiation
Radiation is generally measured in units of rads, or radiation absorbed dose, which is equivalent to 0.1 J of energy emitted per kg of isotope. Another common unit of measure is the rem, or radiation equivalent for mammals, which is equal to a rad in most cases. To give you some qualitative idea, a dental x-ray produces the equivalent of 1 millirem. The natural background level of radiation you receive is approximately 200 to 300 millirem per year, depending on where you live. "Natural background" is composed of radiation from cosmic rays, the ground, bricks, stone in buildings, radon gas, medical procedures, and potassium in your body. The government limit of acceptable radiation for the general public is 500 millirem in a year, not counting what a doctor may prescribe for you. The first medical sign of radiation sickness occurs after a single dose (all at once) of 25 rem (25,000 mrem), although lower levels may increase the risk of developing cancer at some point in life.
Types of Spontaneous Radioactive Decay
Several types of radioactive decay occur to make an unstable nucleus more stable. Alpha emission is the loss of a helium nucleus (2 protons and 2 neutrons) which carries away a mass of four atomic mass units, or amu. The charge of the alpha particle is +2.
| Name | Symbol | Charge | Atomic # | Nuclear loss | Ability to Penetrate |
|---|---|---|---|---|---|
| Alpha | + 2 | 2 | 2 p and 2 n | lowest | |
| Beta | - 1 | 0 | 1 n or 1p | low | |
| Gamma | No charge | 0 | No change | High |
During alpha emission, the atomic mass decreases by four and the atomic number decreases by two. Alpha decay usually occurs in elements with atomic numbers greater than 82 which do not contain enough binding energy to hold together the massive nucleus. A typical alpha emission is the decay of a heavy isotope such as plutonium-239 to uranium- 235.
Excess binding energy is given off by the kinetic energy of the alpha particle and sometimes by the emission of gamma energy. Gamma energy is emitted as photons and is a type of electromagnetic radiation.
Elements below atomic number 40 generally have stable nuclei with an equal number of protons and neutrons (1:1 ratio). As the atomic number increases from 40 to 108, the stable neutron to proton ratio increases toward 1.5 neutrons to 1 proton. Beta decay is the loss of an electron from the nucleus. Usually there are no electrons in the nucleus. During beta decay one of the numerous neutrons changes into a proton and an electron. This electron from the nucleus is called a beta particle and is ejected from the nucleus. During beta decay, the number of neutrons decreases by one and the number of protons increases by one. The atomic mass remains the same. Also note that the overall charge is conserved.
no = p+ + e-
The decay of carbon-14 into nitrogen-14 and a beta particle is an example of this type of decay.
Another form of radioactive decay is positron emission, the loss of a positron (positive electron) from a nucleus that has an excess number of protons. Elements that have a higher proton to neutron ratio than normal can decay by positron emission. Here a proton splits into a neutron and a positron (e+). During positron emission the atomic number decreases by one and the number of neutrons increases by one as a proton is converted to a positron and a neutron. The atomic mass remains the same, and the overall charge is again conserved.
p+ = e+ + no
The decay of carbon-10 to boron-10 is an example of this type of reaction.
Gamma emission refers to the discharge of high-energy electromagnetic radiation from an atom. Energy loss in the form of gamma emission occurs when the nucleus is in an excited state and returns to its ground or normal state by releasing a gamma particle, or high-energy photon. During gamma emission, neither the atomic mass nor the atomic number change.
An example of this reaction is the emission of gamma radiation from barium -137m. The "m" stands for "meta stable," which means it is stable only for a limited time.
Induced Radioactive Decay
The type of radioactive decay which occurs in nuclear reactors is induced by particle bombardment and is called transmutation. We have been able to create hundreds of isotopes and new elements by bombarding existing isotopes with subatomic particles or even the nuclei of light elements. This concept is the basis of fission, splitting of the nucleus; and fusion, joining of two nuclei.
Neutron bombardment is the process of "hitting" a nucleus with a free thermal neutron (one with the correct amount of energy) in order to split it into lighter products. Several products are possible when splitting the nucleus of an atom. A common example is the fission of 235U in a nuclear reactor. Free neutrons are almost always among the products, which propagate the reaction with other nuclei, called a chain reaction. When 235U is bombarded with very low energy free neutrons, a fission reaction occurs where the products may be krypton- 92, barium -141, three neutrons, and possibly gamma radiation.
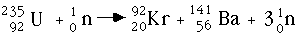
Many different products could have occurred as a result of the breakdown above. The criterion is that the numbers of amu's, protons, and neutrons of the products add up to that of the reactants, much like a chemical reaction. Note that the amu's add up : 235 + 1 = 92 + 141 + 3, as well as the number of protons: 92 + 0 = 36 + 56 + 0 and neutrons: 143 + 1 = 56 + 85 + 3.
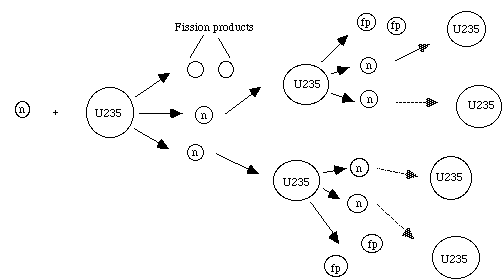
Figure 7: This diagram represents a chain reaction of 235U. A neutron hits the uranium atom which breaks into fission products, and releases 2 neutrons. The new neutrons carry out the chain reaction as shown.
Another way to induce decay is by striking the nucleus with a helium nucleus, or alpha bombardment. When an alpha particle reacts with a nitrogen-14 atom, an oxygen-17 atom forms and the energy is released as a hydrogen-1 atom and gamma radiation.
Bombarding an aluminum-27 atom with an alpha particle produces phosphorus-30 and a neutron.
Proton bombardment is the bombardment of the nucleus with a proton. When lithium-7 reacts with a proton , two helium-4 atoms are produced.
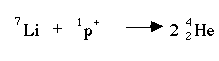
Nuclear Fusion
Another type of induced nuclear reaction is nuclear fusion. Fusion joins together two small nuclei. Fusion reactions supply the power needed for our sun to shine. The principle behind fusion is that in fusing two small nuclei like deuterium (2H, or one neutron and one proton), a great deal of energy is released. Deuterium (2H) and tritium (3H) are both isotopes of hydrogen which are obtainable in normal water. The reaction of deuterium and tritium follows:
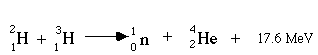
Note that 17.6 million electron volts of energy is released per 4He produced. This is due to the difference in the atomic energies of the reactants and products. Conditions needed for fusion are high concentration of fusing elements, high temperature, and high density. It has been estimated that, reacting only 1 gram of deuterium will release an amount of energy equivalent to 2,400 gallons of gasoline.
Nuclear energy (from fission, not fusion) supplies about 7% of the total energy used in the United States, and 22% of the electrical energy used in the United States in a year. Energy from nuclear reactors is considered to be "clean" energy, as carbon and nitrogen oxides as well as smoke and soot are not released into the atmosphere. The disadvantages of nuclear energy include the high cost of building nuclear power plants, finding a politically acceptable ways to dispose of the radioactive wastes including the spent fuel rods, the risk of radioactive release, and the cost of shutting down a nuclear power plant at the end of its useful life.
Types Of Nuclear Reactors
Nuclear reactors use the process of fission, or atom splitting, to release the energy from the nucleus of the fuel. Typically, 235U is used as fuel in most nuclear power plants because it is a fissile material, one which will undergo fission upon encountering any neutron, especially a very slow one. The added neutron causes the nucleus to become unstable, and it splits into nuclear fragments.
Nuclear reactors use enriched uranium (3% uranium-235 and 97% uranium-238) as the fuel source. Since normal uranium found in the ground is only 0.7% 235U and 99.3% 238U, we put it through an enrichment process in order to increase this percentage to 3%. The fuel is placed in the core of the nuclear reactor. A chain reaction is initiated by bombarding the fuel with slow neutrons because slower neutrons with less thermal energy have approximately 1,000 times greater chance of causing a fission reaction to occur than faster neutrons. Neutrons must be slowed down by water or graphite moderators to produce a chain reaction. The process repeats itself rapidly or slowly depending on the presence of control rods. Control rods are made of cadmium or boron which absorb neutrons to control or stop a chain reaction. Heat from the chain reaction and the resulting speed of the fission products, such as Kr and Ba in the last example, is absorbed by water in the reactor. Very fast neutrons, or thermal neutrons, are released too. If there is no moderator to slow them down, the chain reaction will stop by itself. U.S. reactors use the cooling water as the moderator. If the coolant is lost or the reactor gets too hot and it boils away, the moderator is lost as well, and the reaction stops. Unfortunately, the Russian Chernobyl reactor was designed with a graphite moderator. When the coolant was lost the reactor got even hotter and caught on fire. Because water is both the moderator and the coolant in U.S. reactors, an accident like Chernobyl cannot physically occur.
There are several types of nuclear reactors in use today. The first two types of nuclear reactors we will discuss are light water reactors, or LWRs. LWRs account for about 80% of the world's nuclear power production. One type is the boiling water reactor, or BWR. In a BWR, water is pumped through the reactor core where it serves two functions: moderator and coolant. The thermal energy released by the nuclear reactions is absorbed by the water which turns into steam. The steam is shot through turbines that run generators for electricity production.
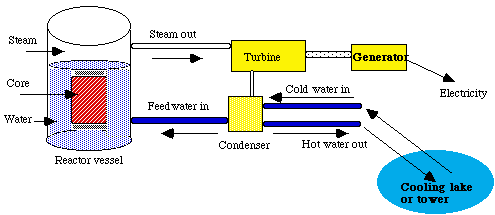
Figure 8: Schematic of a Boiling Water Reactor (BWR). The water in the reactor vessel is heated by the nuclear reaction in the core. The water then turns into steam which is directed to a turbine and then a generator to produce electricity. The hot water then cycles through a condenser which is flooded with cold water from the lake or cooling tower in order to exchange heat. The warm water goes out to the lake and the cool water into the reactor vessel.
The other type of LWR is the pressurized water reactor, or PWR. In a PWR, the water is pressurized to prevent it from boiling, even at temperatures approaching 315o C. The water in the core passes through a primary loop which passes through a heat exchanger. The heat exchanger removes the thermal energy in the primary loop and transfers it to water in a secondary loop. The water in the secondary coil flashes into steam due to the decreased pressure as it is passed through a series of turbines that drive electrical generators. It is important to note that regardless of the fuel, all power plants use steam to drive turbines to generate electricity.
The third type of nuclear reactor is the breeder reactor, so-named because it makes more fissile fuel than it uses. In a breeder reactor, some of the usually nonfissile 238U is converted to 239Pu, which is fissile, by the series of beta decays in the following reaction:
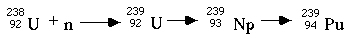
This reaction is facilitated by the use of fast neutrons because they are more likely to be caught by 238U than by 235U. A general nuclear reaction usually releases fast neutrons, therefore, breeder reactors are inherently efficient. They also produce heat and require a coolant. Liquid sodium is commonly used as the heat exchange medium, or coolant, because it does not slow down high speed neutrons like water does.
The main advantage of a breeder reactor is that it generates more nuclear fuel, 239Pu, than it uses. It converts 238U, a plentiful non-fissile, or stable, isotope into a fissile one, making this fuel source virtually inexhaustible. Supplies of 238U will last for more than the next 40,000 years. There are also some disadvantages of breeder reactors. If plutonium escapes into the environment, it carries a high health risk due to its toxicity. Plutonium is also used to make atomic weapons. Though 235U can also be used for weapons, it requires such an enormous amount of effort to purify it that it is not easily used. Breeder reactors also have the ability to melt down if problems occur. This problem has been solved recently by Argone National Laboratory through the development of a Fast Breeder Reactor. The nuclear reaction will stop automatically if the fuel temperature gets to high in this type of reactor. Unfortunately, the U.S. congress has removed funding from this project which held a safe answer to our energy needs.
Currently, the United States does not use breeder reactors to provide nuclear energy. France has used breeder reactors, and Japan is planning to use breeder reactors to generate both electricity and more nuclear fuel.
Nuclear Waste
One obstacle to using nuclear power is that the nuclear waste generated cannot just be thrown in a dumpster, it is radioactive! High-level radioactive waste, the fission products in the spent fuel rods, will be dangerous for the next hundred or thousand years. Engineers have developed ways of storing this waste in hopes of protecting the environment. Spent fuel rods are first stored in large tanks or swimming pools on the site of the power plant to remove the heat left over from the reaction. Once they are no longer thermally hot, the spent fuel rods, usually in the form of small metal tubes, are encapsulated in ceramic or glass containers which can withstand radioactive decay. These small containers are then placed in stainless steel containers which are stored underground in large caves. Because the waste is all in solid form, nothing can leak from the inside. In order to prevent water from leaking in, materials are placed all around the waste which will absorb any ground water that may seep in. These "caves" are always contained within very stable geologic formations. This type of containment system is called a multiple barrier containment system.
Fortunately, very little high level waste is made per reactor per year. Unlike a coal plant which produces about 15 tons of carbon dioxide, 200 pounds of sulfur dioxide, and about 1,000 tons of solid ash per minute, the high level waste from one year of nuclear power plant operation produces about 1.5 tons and would occupy a volume of about half a cubic yard, which could easily fit under your coffee table!
However, other things become radioactive in the process of operating a nuclear power plant. Objects like water and air filters for trapping radioactive material, rags, gloves, lab equipment, pipes, and mops are considered low-level radioactive waste. They have been used near or in the reactor and were exposed to neutrons. About 25% of all low-level waste comes from hospitals, research labs, and industry. Although the radioactivity in low-level waste is about a million times lower than that in high level waste, it occupies about 1,000 times the volume of high-level waste. Because the radioactivity is so low, low-level waste is buried at about 20 feet underground in controlled areas and allowed to decay.
A major problem with radioactive waste is not the amount or even what to do with it. The real problem is the public's perception. Most people seem to agree that we need to do something with this waste, but no one wants it in their neighborhood, no matter how safe the containment structure. Here is another instance where energy becomes perhaps a greater political issue than a scientific one.
Catastrophic Accidents
One major event in the history of American nuclear reactors was the accident at the nuclear power plant, a PWR, at Three Mile Island in 1979. Many term the accident a "near meltdown," while others feel that this is a gross over-exaggeration. The cause of the accident was a failed valve, which allowed water to run out of the pressure vessel. Of course, with no moderator, the chain reaction stopped. However, the core was still hot and needed to be cooled to keep the fuel from melting and ruining the reactor. The engineers thought the valve was closed due to a faulty indicator on their instrument panel, but were able to determine the cause of the problem and close an auxiliary valve to keep the water from escaping. Had the water continued to run out for 30-60 more minutes, the loss of coolant may have caused a meltdown. A meltdown occurs when the fuel rods become so hot that they melt, allowing the radioactivity to escape into the reactor vessel. However, what many people don't realize is that the containment structure for the reactor is designed to keep all the radioactivity inside and filter it out of the inside atmosphere in the event of an accident. Containment structures are tested for susceptibility to tornadoes, earthquakes, airplanes flying into them (really), and explosives. In the case of Three Mile Island, the containment structure worked and very little radioactive material was released into the environment.
The world's most serious nuclear power plant accident occurred in 1986 when the plant at Chernobyl, Russia exploded. The Chernobyl reactor is an RMBK type reactor, which uses water as the coolant and graphite as the moderator with natural uranium. As a result of a poorly conducted test, the coolant ended up at a low level in the reactor vessel, which prompted the removal of some control rods. When the power increased back to normal levels, there was not enough time to replace them. Thus, the coolant was at a very low level and began to boil. Due to the reactor design, the graphite moderator was still intact, and the chain reaction continued. The high temperature and heat built up and caused two chemical explosions (like dynamite), not nuclear ones, which blew off part of the top of the reactor building. Note that there was not a sophisticated containment structure surrounding the reactor vessel like those in the United States. As a result, a large amount of the radioactivity that was once in the reactor core was dispersed to the surrounding areas as radioactive dust. The fires were eventually put out by dropping sand onto the reactor by helicopter.
Common Questions And Misconceptions
Here are a few common statements nuclear power and responses to them.
S. Nuclear power plants emit radioactivity.
R. False. The radioactive material used in nuclear power facilities is contained in the fuel rods in the core of the reactor. In some reactors, the water coolant also becomes slightly radioactive, but has a short half life and is also contained inside.
S. Nuclear power is dangerous because it cannot be controlled.
R. False. Control rods are kept in the core of reactors with elements (cadmium or boron) which absorb the free neutrons that propagate chain reactions. Also, if the water boils away in LWR's (the U.S. and European reactors) the reaction stops because the moderator is missing.
S. Nuclear power plants are environmentally unsound because of the waste heat released into the environment by the water.
R. True. The hot water released from the reactors (which is often stored in ponds near the reactor) does release more heat into the environment, which could cause problems in the immediate environment of the plant. However, fossil fuel power plants release about twice as much heated water for making the same amount of electricity and therefore have the same environmental problems. Cooling towers can alleviate this.
S. Nuclear power avoids several of the environmental problems associated with fossil fuels.
R. True. Because nuclear reactions are not based on combustion, greenhouse gases are not released, and nuclear power plants are therefore not a contributor to the greenhouse effect. Similarly, there is no harmful SO2 emission that causes acid rain. Nuclear power plants also tend to require less land than fossil fuel plants which decreases the impact on the immediate environment of the plant.
S. A nuclear power plant is unsafe because it can explode like a bomb.
R. False. An atomic bomb is composed of extremely concentrated 235U and 239Pu. Explosives force them together, and the chain reaction proceeds so fast that an extremely large amount of energy is released in a very short time. In a nuclear reactor, the percentage of 235U in enriched fuel is only 3%, as opposed to 95% in a bomb. Thus, it is physically impossible for a nuclear power plant to explode like a nuclear bomb.
S. There is so much radioactive waste generated that it is inefficient to utilize nuclear power for a large portion of our energy.
R. False. The amount of high-level radioactive waste generated by a power plant to produce the amount of energy a person will use over a 70 year lifetime is about the size of a soda can. Compare that to the tons of ash produced by a coal power plant! Low level wastes increase the amount slightly, but they generally have short half lives and can be stored economically until they can be safely disposed of.
S. High level waste can not be cleaned up.
R. True. There is no known technologically feasible or biological means to quickly remove the radioactivity from high level waste.
S. Nuclear power is very controversial and right now this controversy can not be resolved.
R. True. The good and bad issues associated with nuclear power make it difficult for anyone to say it should or should not be used. Until better means of disposing or making less waste are found, some people will be against this energy form.